Retatrutide (10mg Vial)
Retatrutide is a synthetic peptide currently under investigation in preclinical and early clinical research settings. It is classified as a multi-receptor agonist (GLP-1, GIP, and glucagon receptors) and is being studied for its potential metabolic effects.
Available information is derived primarily from laboratory research, animal studies, and limited early-phase human trials. Long-term safety, efficacy, and appropriate use parameters have not been established.
Retatrutide is not approved by the FDA or any global regulatory authority for medical use. All information presented below is provided strictly for educational and scientific discussion purposes.
⸻
“Reconstitution” is a fancy word that just means:
Mixing powder with water so it becomes liquid
In research settings:
- Scientists add special clean water (BACwater) to the vial
- The powder slowly melts into the water
- They wait a bit so it mixes evenly
Reconstitution Reference (Laboratory Context Only)
The following reflects commonly cited laboratory handling references and does not imply approval or recommended use:
•Vial content: 10 mg lyophilized Retatrutide
•A commonly referenced reconstitution volume is 2.0 mL of bacteriostatic water (BAC water) added using sterile laboratory technique.
•With a standard U-100 insulin syringe, 2.0 mL equals two full syringe draws to the “100” unit mark.
•After reconstitution, the vial is typically allowed to sit undisturbed for approximately 20 minutes to ensure full dissolution.
Resulting concentration:
10 mg ÷ 2.0 mL = 5 mg per mL
⸻
Referenced Research Amounts (Preclinical / Early Clinical Context)
•Research discussions commonly reference a typical weekly range of 2 mg to 12 mg, administered once weekly.
•Gradual increases from lower referenced amounts are often noted in non-clinical discussions.
•These values are not approved dosing recommendations and are derived from investigational and early-stage research data only.
Note: A single 10 mg vial cannot supply more than 10 mg total. Any reference above this amount is theoretical only and shown strictly for mathematical illustration.
⸻
Volume Conversion Reference (U-100 Syringe)
The table below reflects mathematical volume conversions only based on a 10 mg vial reconstituted with 2.0 mL BAC water (5 mg/mL concentration).
This table does not imply safety, efficacy, or appropriate use.
Conversion Table (10 mg vial + 2.0 mL dilution)
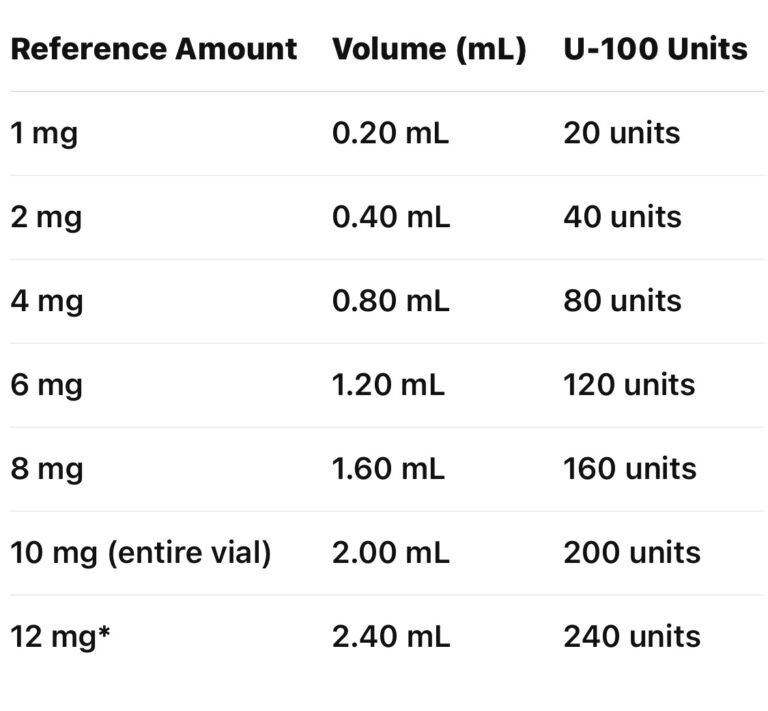
*Exceeds the contents of a single 10 mg vial; shown for mathematical reference only.
Example (Mathematical Only):
A 2 mg reference amount corresponds to 0.40 mL, which equals 40 units on a U-100 syringe.
2mg = 40 units on a U-100 syringe
⸻
Regulatory & Use Disclaimer
•This content is provided solely for educational, academic, and scientific research discussion.
•It does not constitute medical advice, diagnosis, or treatment.
•Retatrutide is investigational and lacks sufficient long-term human clinical data.
•Not approved for human or veterinary use.
•Not for human consumption.
•Any handling or study should occur only in appropriately authorized research settings and in compliance with all applicable laws and regulations.
Quick Recap
