Tesamorelin is a synthetic 44-amino-acid analog of Growth Hormone–Releasing Hormone (GHRH).
Mechanism of action
•Binds to pituitary GHRH receptors
•Stimulates pulsatile endogenous growth hormone (GH) release
•Increases IGF-1 levels
•Leads to:
•Increased lipolysis (fat breakdown)
•Improved metabolic signaling
•Protein synthesis effects
Studied / clinical effects
•Reduction of visceral adipose tissue (VAT)
•Improvements in lipid profiles
•Potential reduction of liver fat (NAFLD research)
•Investigational cognitive and aging research via GH/IGF-1 restoration
Regulatory status clarification
•FDA-approved as a prescription medication for reduction of excess abdominal fat in HIV-associated lipodystrophy.
•Some research-grade products sold online are labeled “research only,” but that refers to vendor classification — not the molecule itself.
⸻
Reconstitution math (educational reference)
You described:
•Vial: 10 mg tesamorelin
•Added: 2.0 mL bacteriostatic water
Resulting concentration
Final solution concentration:
 5 mg per mL
5 mg per mL
In clinical trials and approved therapeutic use:
 Common studied dose:
Common studied dose:
•2 mg once daily (subcutaneous)
(This is the most widely used standardized dose in HIV lipodystrophy trials and many research protocols.)
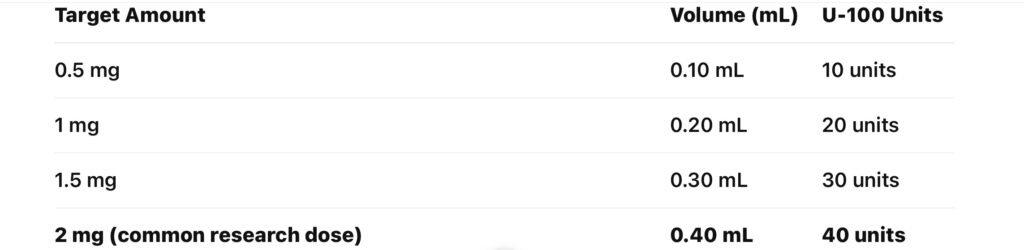
Average research dose marking:
40 units on a U-100 syringe
In human studies, protocols commonly include:
•Subcutaneous administration
•Daily dosing schedule
•Injection site rotation (abdomen commonly used in trials)
•Monitoring endpoints:
•IGF-1 levels
•Body composition (DEXA/CT)
•Lipid profile
•Glucose metabolism
•Duration:
•Many trials: 6–12 months
⸻
 Important safety notes
Important safety notes
•GH-axis peptides can affect glucose regulation, fluid balance, and IGF-1 levels.
•Actual clinical use should be supervised by a licensed medical professional.
•Research-grade compounds may differ in purity or sterility from pharmaceutical products.